NEWS & AWARDS
ALL SYNAPSY'S NEWS
SEARCH SYNAPSY’S NEWS
JUMP TO A SPECIFIC MONTH
The future of psychiatric neuroscience is in sight
30 September 2022
Twelve years after its creation, Synapsy closes the NCCR chapter and opens a network of faculty Centers in psychiatric neurosciences. The NCCR Synapsy came to an end on September 30th,...
Read More
Synapsy at the heart of Campus
29 September 2022
The scientific magazine of the University of Geneva, Campus, has dedicated its 150th issue to the NCCR-Synapsy. Campus reviews Synapsy’s major contributions to the early identification of symptoms of psychiatric diseases. It also highlights...
Read More
Schizophrenia: a clear view of the world
29 September 2022
A study by EPFL and Synapsy has shown that schizophrenia does not impact the brain's ability to assemble and interpret images – it just affects the speed at which complex...
Read More
Closing events: cultivate the link with society
30 June 2022
On May 12 and June 2 2022, the NCCR-Synapsy celebrated 12 years of research in psychiatric neuroscience, synonymous with the end of the support of the Swiss National Science Foundation,...
Read More
A forward-looking poster session!
14 April 2022
The last NCCR Synapsy retreat, held on April 4 and 5 in Les Diablerets, was a time of emotion, sharing and excellence! The pooling of researchers, fuelled by clinical psychiatry...
Read More
Social interactions divert the established motivational system
31 March 2022
A Synapsy study has demonstrated that the first step required to embark on a social interaction, moving towards others, follows a neural pathway that lies outside the system of motivation...
Read More
Post-traumatic pain expressed physically from generation to generation
29 March 2022
A Synapsy article has demonstrated that mothers suffering from post-traumatic stress disorders resulting from domestic violence often convert their mental suffering into physical pain and pass on this somatisation to...
Read More
Get More or Give Up, a Question of Anxiety
23 March 2022
Individuals can respond to stress in diametrically opposed ways. A NCCR-Synapsy study shows that the behavioral response depends on the level of anxiety and identifies the cerebral mechanisms involved. People...
Read More
Connecting stress, weight, and social anxiety in early adolescence
9 March 2022
Scientists at EPFL have found a biological connection explaining why there is an increased predisposition to develop obesity and being less sociable in individuals that have experienced stress during early...
Read More
Communication defect in psychotic disorders
3 March 2022
Scientists at the UNIGE demonstrate how a defect in communication between brain areas is linked to the onset of psychotic disorders. Communication between brain areas is crucial for the brain to...
Read More
Recruiting participants for a study on brain metabolism and behavior
3 February 2022
The Laboratory of Behavioral Genetics (LGC) of the Brain Mind Institute at EPFL is looking for participants for a study on the brain basis of behavior. If you are interested, we...
Read More
A mother’s lack of empathy impacts her child’s mental health
1 February 2022
A Synapsy study shows that mothers with post-traumatic stress disorder due to interpersonal violence misread their children's emotions due to a lack of emotional comprehension, a difficulty that, in turn,...
Read More
Gene-environment interactions that drives autism
31 January 2022
A UNIGE team demonstrates how genes and environment interact in autistic disorders, thus explaining the vast variability of the disease’s symptoms. People with autism spectrum disorders all have characteristic behavioural difficulties. Nevertheless,...
Read More
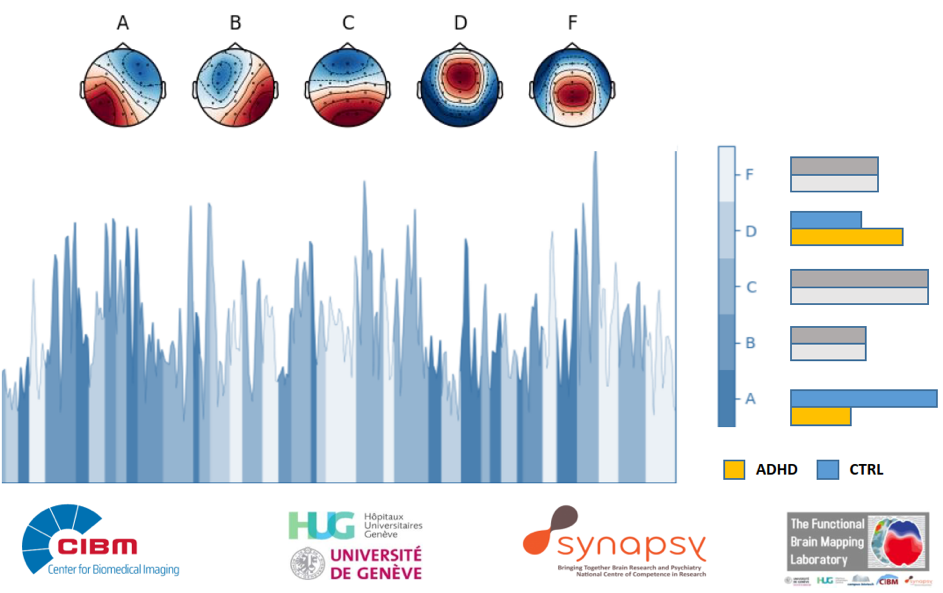
Detecting Attention Deficit Disorder More Accurately
6 January 2022
By analysing the fluctuations of different cerebral states, neuroscientists at Synapsy have identified a new neurological marker for attention deficit disorder with or without hyperactivity.Adult diagnosis of attention deficit hyperactive...
Read More
Le Podcast « Toi aussi ? » tend le micro aux jeunes pour parler de santé mentale
17 December 2021
Depuis bientôt deux ans et la survenue de la pandémie, le sujet de la santé mentale s’impose dans le débat public. L’isolement, le manque de perspectives et les mesures inhibant...
Read More
Two blood markers for schizophrenia
16 December 2021
A research team from Synapsy has shown that the severity of the clinical symptoms of schizophrenia is strongly linked to blood biomarkers related to the deregulation of neuronal mitochondria.Psychotic symptoms...
Read More
Tracking neurons that make us sociable
2 December 2021
A team from Synapsy has discovered that neurons linked to the reward system are responsible for motivating us to interact with our fellow human beings. Human beings, like most mammals,...
Read More
Same signature for several mental illnesses
23 November 2021
A team from NCCR-Synapsy has identified a common neurobiological signature of dysfunction in the brain circuits involved in regulating emotions in three clinically-distinct psychiatric illnesses.People suffering from attention-deficit hyperactivity disorder...
Read More
Women researchers win Synapsy poster awards
17 November 2021
The poster session of the eleventh annual meeting of the NCCR-Synapsy took place at the Diabrlerets Convention Center on the evening of Thursday, October 14. Peter Uhlhaas, representative of the...
Read More