iTango: A switch to illuminate the neurons
3 April 2017
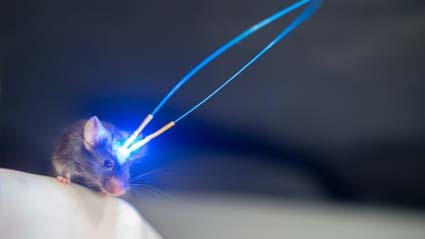
The blue light activates the iTango system. Mouse implanted with an optical fiber that directs light to the parts of the brain involved in the addictive behavior.
Swiss and American researchers are developing a technique of unprecedented precision to visualize and manipulate the action of neurotransmitters.
The transmission of information in the brain goes through neurotransmitter molecules which diffuse themselves through the junction zone between two nerve cells called the synapse. In some cases, these molecules can spread into the tissue, thus inundating different types of nerve cells, it is called “neuromodulation”. To better understand the impact of neuromodulators on cerebral circuits and behavior, it is necessary to be able to identify the stimulated neurons and then to follow their activity.
A collaboration between a team from the University of Geneva (UNIGE) and the Max Planck Institute for Neurosciences (MPFI) in Florida has solved this problem with a new technique called iTango, which makes it possible to control in real time the cells subjected to the neuromodulation. iTango is based on an innovative gene expression system based on light, it will allow scientists to better understand the mechanisms of control of the brain circuits involved, for example, in addiction or in certain psychiatric disorders such as schizophrenia. Results are published in Nature Methods.
The Tango system is a technique developed almost 10 years ago to observe individual neurons under the effect of neuromodulators. Once stimulated, these neurons express a green fluorescent protein that allows researchers to identify them. But the system encounters a major defect: when the protein sensors are overexpressed, they produce this fluorescence continuously. More and more neurons become fluorescent, preventing to distinguish the populations of neurons actually stimulated at a given moment, or during a specific behavior of the animal. This technique was therefore little used to study neuromodulation states in mammals. “If Tango had its limits, the concept itself was interesting. That’s why we wanted to make it more effective by adding a kind of switch, “explains Hyungbae Kwon, neuroscientist at MPFI and co-author of this work. With iTango – for “Inducible Tango Technique” – the cells become fluorescent only if two conditions are met. On the one hand, a particular neuromodulator must be present (which can change according to the research subject) and, on the other hand, the researchers must diffuse a specific blue light on the neurons they want to study. If they turn off the light, the protein stops shining. The addition of this switch makes it possible to precisely control the fluorescence and thus to identify the neuronal populations concerned much more easily.
A technique applicable to many fields
To test their technology, the researchers used iTango to identify neuromodulatory dopamine-activated neurons in particular behaviors in mice. They were able to identify two populations of neurons – one involved in increased locomotion and the other in the sense of reward. By carrying on their work a little further, they have been able to inhibit, or rather induce, behaviors associated with the neuronal populations that they had previously identified. “These behaviors are typically seen in individuals who have consumed cocaine,” says Prof. Christian Lüscher of the UNIGE Faculty of Medicine, who co-led this study. “This shows iTango’s interest in understanding how dopamine controls the circuitry of motivation and ultimately addiction. The use of light as a switch also offers a huge advantage: this technique can be applied to almost any type of cell signaling. “ITango will therefore be useful in many fields: neuropsychic diseases, of course, but also the development of drugs in targeted therapies against cancer. Indeed, one of the molecules that make up the iTango system is a sensor of the G protein, a multifunctional protein involved in the transfer of information inside the cells. A revolutionary technique, therefore, in many fields of research in the life sciences.
Author : UNIGE (FR) >
Translation : Tania Secalin
Reference :
Lee D1,2, Creed M3, Jung K1, Stefanelli T3, Wendler DJ1, Oh WC1, Mignocchi NL1, Lüscher C3,4, Kwon HB1,5;
Temporally precise labeling and control of neuromodulatory circuits in the mammalian brain.
Nature Methods 14(5):495-503, May 2017. doi:10.1038/nmeth.4234 >
